News you can use from Pepperdine's Student Health Center. Stay up to date on the latest happenings and health-related announcements.
Expected Fall 2022 COVID-19 Prevention Measures
As the community prepares for the 2022–2023 academic year, Pepperdine continues to prioritize the health and safety of our community. For your awareness and planning, the University is sharing the following overview of COVID-19 protocols currently in effect and expected for fall 2022.
Get a COVID-19 Vaccine Booster Shot and Upload Documentation
Students who receive a COVID-19 vaccine booster shot are asked to upload a photo, scan, or screenshot of their booster dose to their Patient Portal. Individuals who are up to date on all COVID-19 vaccines are not required to quarantine if they have been in close contact with someone with COVID-19.
Get the Most out of Masking
Correct and consistent mask use is a critical step everyone can take to prevent getting and spreading COVID-19. This is more important than ever with new, more easily transmitted variants of COVID-19 on the rise.
Spring 2022 COVID-19 Testing Requirements
All students are required to test negative within 72 hours prior to returning to any Pepperdine campus. Additionally, in light of the current trends every student must—prior to attending a class in person at a Southern California campus—also receive a negative test result from a Pepperdine screening testing site.
Get Your COVID-19 Booster Shot
COVID-19 vaccines are effective at protecting you from getting sick. Based on what we know about COVID-19 vaccines, and similar to what we know from other recommended vaccines, people who are up to date with their vaccines are well protected from serious illness or other health outcomes.
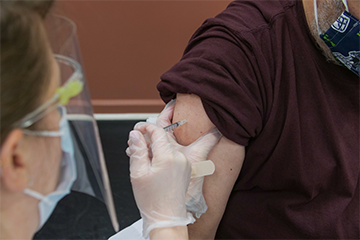
Upcoming COVID-19 Booster Vaccine Clinics
Pepperdine, in partnership with Rite Aid, is happy to offer COVID-19 booster vaccination clinics for all faculty, staff, students, and their eligible family members including spouses and children.
COVID-19 Vaccine Clinics
The Student Health Center offers first and second doses of Moderna and Pfizer COVID-19 vaccines at no cost to Pepperdine community members. Vaccine clinics are held weekly every Thursday at the Student Health Center on the Malibu campus. Schedule your appointment today by calling 310.506.4316 and selecting option 3 when prompted.
Who is Eligible for a COVID-19 Vaccine Booster Shot?
COVID-19 vaccine booster shots are available for certain Pfizer-BioNTech vaccine recipients who completed their initial series at least 6 months ago.
COVID-19 Vaccines for Moderately to Severely Immunocompromised People
CDC recommends that people with moderately to severely compromised immune systems receive an additional dose of mRNA COVID-19 vaccine at least 28 days after a second dose of Pfizer-BioNTech COVID-19 vaccineor Moderna COVID-19 Vaccine.
Vaccination Verification Requirements to Begin at Outdoor Mega Events and Indoor Portions of Bars, Lounges, Nightclubs, Breweries, Wineries, and Distilleries
Beginning Thursday, October 7, proof of full vaccination or a negative test result will be required to enter outdoor mega events, and proof of at least one dose of the COVID-19 vaccine will be required to enter or work in indoor portions of bars, lounges, nightclubs, breweries, wineries, and distilleries in L.A. County.